A pullet that reaches the laying house without adequate immune protection is not a productive asset. She is a liability — one disease challenge away from a mortality event, a production dip, or a subclinical immune suppression that depresses laying rate for months without ever producing a diagnosable outbreak.
Vaccination is not the most visible part of pullet management. It does not show up in daily feed records or weekly weight data. But its effect on 52-week production performance is as measurable as nutrition or ventilation — and its failures are far more expensive to diagnose, because they look like everything else.
A Newcastle disease outbreak in a vaccinated flock looks like a vaccination failure. It might be. It might also be a cold chain breakdown, an incorrect dilution, a wrong administration route, poor maternal antibody mapping before the vaccination schedule was designed, or a vaccine strain mismatch with the circulating field strain. Understanding which failure occurred requires knowing how the vaccination program was constructed and why each decision was made.
This guide builds that understanding: the disease threats that matter in West and Central Africa, the immunological logic behind each vaccine and its timing, the administration requirements that determine whether a vaccine works or fails, and the cold chain disciplines that separate a vaccination program that protects from one that only appears to.
Why Timing Is Everything
Before covering the schedule, the biology behind it must be established. Vaccination timing is not arbitrary. It is calibrated to three overlapping immunological windows that determine when a vaccine can produce protective immunity — and when it cannot.
Maternal Antibody Interference
Day-old chicks carry passive immunity transferred from the hen through the yolk. These maternal antibodies provide early protection against pathogens the hen was vaccinated against or exposed to, but they also interfere with live vaccine replication. A live Newcastle disease vaccine administered at day 1 in a chick with high maternal antibody titers against Newcastle disease will be neutralized before it can stimulate active immunity. The vaccine appears to have been given. No protective titer is produced.
Maternal antibody levels vary between flocks and between individual birds within a flock, depending on the vaccination history and health status of the parent flock. Maternal antibody half-life for Newcastle disease is approximately 4–5 days; for Gumboro (IBD), approximately 3–4 days. By day 10–14, most maternal antibody protection has waned to levels that no longer interfere with live vaccine replication but also no longer provide protective immunity against field challenge.
This is why the first Newcastle disease vaccination is typically scheduled at day 7–10 rather than day 1: maternal antibodies have partially declined, interference is reduced, and the chick’s own immune system has matured sufficiently to respond to vaccine antigen.
Where parent flock vaccination records are unavailable — common in commercial hatcheries supplying multiple layer operations in West Africa — maternal antibody mapping (ELISA testing of a sample of day-old chicks from each hatch) should be conducted at the start of each new flock to calibrate vaccination timing to actual antibody levels rather than assumed ones.
The Immunological Gap
Between the decline of maternal antibody protection and the establishment of vaccine-induced active immunity, there is a window of susceptibility — the immunological gap — during which the bird has neither passive nor active protection. This gap is most dangerous for Gumboro disease (IBD), where the bursa of Fabricius is both the target organ for the pathogen and the site of B-lymphocyte development. A Gumboro infection during the immunological gap destroys the bursa — the immune factory — before the immune system has produced enough B-cells to defend it. The result is immunosuppression that is permanent within that production cycle and leaves the bird unable to respond adequately to any subsequent vaccination.
Correct Gumboro vaccination timing — bridging the maternal antibody window to active immunity without leaving a gap — is the most technically demanding element of the pullet vaccination program.
Vaccine-Induced Immunity Development
Live attenuated vaccines replicate in the host, producing a mild, controlled infection that stimulates both humoral immunity (antibody production by B-lymphocytes) and cell-mediated immunity (T-lymphocyte activation). Protective immunity after live vaccination typically develops within 7–14 days of administration. This development period is why booster vaccinations are scheduled 2–3 weeks after primary vaccinations — the booster is administered when the primary immune response has peaked, and it drives a secondary (anamnestic) response that is faster and produces higher, longer-lasting antibody titers.
Inactivated (killed) vaccines do not replicate in the host. They stimulate only humoral immunity and require an adjuvant to extend antigen presentation. They produce lower and shorter-lasting immunity when given alone, which is why inactivated vaccines are almost always used as boosters after priming with live vaccines — they amplify an established immune response rather than initiating one from zero.
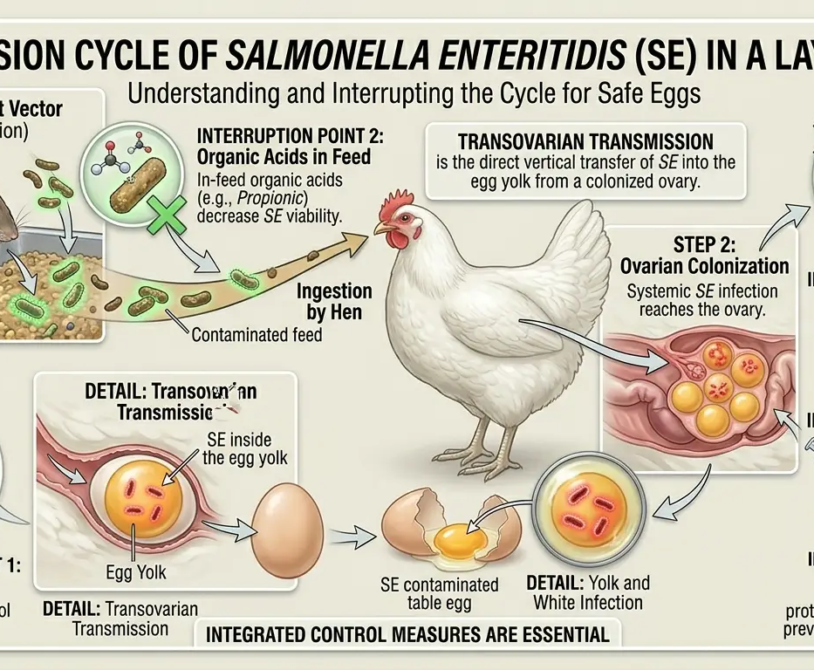
The Disease Priorities: What This Schedule Protects Against
Not every vaccine listed in a textbook is relevant to every production context. The following diseases represent the primary vaccination targets for commercial layer pullets in West and Central Africa, ranked by production impact and field prevalence.
Newcastle Disease (ND)
Newcastle disease virus (NDV) is the single highest-priority pathogen for layer operations across sub-Saharan Africa. Velogenic strains circulating in the region produce mortality rates of 80–100% in unvaccinated flocks. Even in vaccinated flocks, challenge from virulent field strains can cause production drops of 20–40% and egg quality failures that persist for 4–8 weeks after the acute outbreak resolves.
NDV is enveloped and environmentally fragile — it survives hours to days outside the host — but spreads rapidly through respiratory secretions, fecal contamination, contaminated equipment, and movement of people between farms. Its prevalence in smallholder and backyard poultry across West Africa means commercial operations face continuous background exposure regardless of biosecurity level.
Newcastle disease vaccination requires multiple doses across the rearing period, a booster before transfer to the laying house, and ongoing revaccination every 8–12 weeks during lay.
Infectious Bursal Disease (Gumboro / IBD)
Gumboro disease destroys the bursa of Fabricius in young chicks, causing primary immunosuppression that compromises response to all subsequent vaccinations and increases susceptibility to secondary bacterial pathogens, including E. coli, Clostridium perfringens, and Staphylococcus aureus. Subclinical IBD — infection without visible clinical signs — is more damaging in the long term than acute IBD because it suppresses immunity without triggering investigation or treatment.
Infectious Bronchitis (IB)
Infectious bronchitis virus (IBV) is a coronavirus that targets the respiratory tract and, in laying hens, the oviduct. Oviduct damage from early IBV infection — including infection during rearing — produces permanent impairment of egg formation, causing the classic “false layer” condition where the oviduct cannot form a shell around the ovum. False layers occupy space in the laying house, consume feed, and produce no eggs — an invisible productivity drain that standard production records may not separately account for.
IBV has many serotypes with limited cross-protection between them. Strain selection for vaccination must match the serotypes circulating in the local region. Using a Massachusetts strain vaccine where a variant strain (793B, QX, or a local African variant) is the field challenge provides incomplete or no protection.
Marek’s Disease (MD)
Marek’s disease virus (MDV) is an oncogenic herpesvirus that causes lymphoma in lymphoid tissues, peripheral nerves, and visceral organs. Clinical signs — leg paralysis, gray eye, visceral tumors — appear weeks to months after infection. Infection occurs in the first days to weeks of life through inhalation of feather follicle dust shed by infected birds. Once infected, a bird remains infected permanently.
Marek’s disease vaccination is administered at the hatchery on day 1 — the only window before field exposure begins. It is a cell-associated vaccine that must be transported and administered under strict cold chain conditions; unlike most other poultry vaccines, it cannot be freeze-dried and reconstituted — it is supplied in liquid nitrogen dewars or frozen ampules that must be handled precisely.
Fowl Pox (FP)
Fowl pox is transmitted mechanically by mosquitoes and direct contact. Wet pox (diphtheritic form) — affecting the mucous membranes of the trachea and oropharynx — is more severe than dry pox (skin lesions) and can cause respiratory distress, feed intake reduction, and mortality in severe outbreaks. In tropical West Africa, where mosquito pressure is year-round, fowl pox vaccination during rearing provides protection through the productive life of the flock.
Infectious Coryza (Avibacterium paragallinarum)
Infectious coryza is a bacterial disease — not a viral one — but it is vaccine-preventable and highly prevalent in layer operations across West and Central Africa. It causes facial swelling, nasal discharge, reduced feed intake, and production drops of 10–40% in laying flocks. Once introduced to a farm, it persists in the environment and in carrier birds, making vaccination the primary control measure. Bacterin vaccines (killed whole-cell preparations) are used; they require two doses for primary immunity and annual boosters in the laying house.
The Complete Vaccination Schedule: Weeks 1–18
The schedule below reflects the core vaccination program for commercial layer pullets in West and Central Africa. Adapt timing based on maternal antibody data, regional disease prevalence, and veterinary guidance specific to your production context.
| Age | Vaccine | Disease | Vaccine Type | Route | Notes |
|---|---|---|---|---|---|
| Day 1 (hatchery) | HVT ± SB-1 or Rispens | Marek’s Disease | Live, cell-associated | Subcutaneous injection | Must be administered at the hatchery; cold chain critical |
| Day 7–10 | La Sota or Clone 30 | Newcastle Disease | Live attenuated | Eye drop | Preferred over drinking water at this age for dose accuracy |
| Day 14–16 | Intermediate IBD strain (e.g., Bursine-2, Winterfield 2512) | Gumboro (IBD) | Live attenuated | Drinking water | Time based on maternal antibody decline; use an intermediate strain where maternal titers are moderate |
| Day 21–24 | La Sota or Clone 30 | Newcastle Disease (booster) | Live attenuated | Eye drop or drinking water | The booster drives secondary immune response; do not skip |
| Day 24–28 | Intermediate IBD (booster) or Intermediate-Plus strain | Gumboro (IBD booster) | Live attenuated | Drinking water | Use intermediate-plus strain if high maternal titers are suspected or if previous IBD exposure is confirmed on the farm. |
| Week 6 | Fowl Pox | Fowl Pox | Live attenuated | Wing web stab | Verify take reaction at day 7 post-vaccination (small scab at stab site confirms successful vaccination) |
| Week 8 | La Sota or Clone 30 | Newcastle Disease | Live attenuated | Drinking water | The third ND vaccination maintains immunity through the rearing period |
| Week 10–12 | Infectious Coryza Bacterin (Dose 1) | Infectious Coryza | Inactivated bacterin | Subcutaneous or intramuscular injection | Two-dose primary series required; first dose at week 10–12 |
| Week 12–14 | Infectious Bronchitis | Infectious Bronchitis | Live attenuated (Ma5, 4/91, or local variant) | Eye drop or spray | Strain selection critical; match to local circulating serotype |
| Week 14–16 | Infectious Coryza Bacterin (Dose 2) | Infectious Coryza | Inactivated bacterin | Subcutaneous or intramuscular injection | Second dose 3–4 weeks after first; completes primary series |
| Week 16 | La Sota or Clone 30 | Newcastle Disease | Live attenuated | Drinking water | Fourth ND vaccination; final live dose before transfer |
| Week 17–18 | ND + IB + EDS Multivalent Killed Vaccine | Newcastle, IB, Egg Drop Syndrome | Inactivated, oil-emulsion adjuvant | Intramuscular injection | Pre-transfer inactivated booster; amplifies rearing immunity and protects through early lay. |
Administration Routes: Why They Are Not Interchangeable
The route of vaccine administration is not a logistical preference. It determines where in the immune system the vaccine antigen is first presented and, therefore, the type and strength of the immune response produced.
Eye Drop
Eye drop administration delivers vaccine directly to the Harderian gland — a lymphoid tissue behind the eye — and to the mucosal surface of the conjunctiva. Both are primary sites of local mucosal immune response (secretory IgA production) and systemic immune priming. For respiratory pathogens like NDV and IBV, eye drop administration produces more robust mucosal immunity in the upper respiratory tract than drinking water vaccination — the route through which field challenge most commonly enters.
Eye drop vaccination requires individual bird handling. It is labor-intensive but more accurate in terms of dose delivery than drinking water vaccination. At early ages (day 7–21), when accurate dosing is critical and maternal antibody interference must be overcome, an eye drop is preferred for NDV.
Drinking Water
Drinking water vaccination is the practical route for mass administration to large flocks once birds are large enough to drink reliably from drinkers. It requires withholding water for 1–2 hours before vaccination to ensure birds are thirsty and drink quickly, complete consumption of the vaccine solution within 1–2 hours of preparation, protection of the vaccine solution from chlorinated water (use skim milk at 2–4 g/liter to neutralize residual chlorine and stabilize the vaccine), and drinker equipment cleaned of detergent, disinfectant, or metal residues that inactivate live vaccine.
The critical limitation of drinking water vaccination is dose uncertainty. Birds that drink less than the target volume receive an underdose. In a flock with uneven drinker access — water pressure variation, inadequate drinker space — a proportion of birds reliably receive an inadequate vaccine dose every time drinking water vaccination is used.
Wing Web Stab
Wing web stab delivers fowl pox vaccine by inoculating the avascular membrane of the wing web with a two-pronged applicator needle dipped in the reconstituted vaccine. A successful take is confirmed 5–7 days post-vaccination by the presence of a small scab at the stab site. Check a minimum of 20 birds per house at the post-vaccination inspection. A take rate below 90% indicates vaccine failure — either from incorrect technique, compromised vaccine viability, or birds already immune from prior exposure.
Subcutaneous and Intramuscular Injection
Injection routes are used for inactivated vaccines (Marek’s disease HVT, Newcastle disease + IB + EDS killed multivalent, infectious coryza bacterin), where systemic antibody production is the target rather than mucosal priming. Oil-emulsion adjuvanted inactivated vaccines produce depot reactions at the injection site — a visible lump that releases antigen slowly over several weeks, sustaining antibody stimulation beyond the initial dose. Injection site reactions that are excessively large, necrotic, or persist beyond 4 weeks post-vaccination indicate incorrect injection technique (intramuscular injection of an oil-emulsion vaccine intended for subcutaneous use, or injection into scar tissue from a previous dose).
Cold Chain Management: The Most Common Point of Failure
A vaccination program is only as strong as its cold chain. A vaccine that reached the farm correctly but was stored at 30°C for four hours before use may produce zero protective immunity regardless of how correctly it was administered. The bird receives the vaccine. The bird receives no protection. The production data does not distinguish this from an unvaccinated bird until the disease outbreak occurs.
Storage Requirements by Vaccine Type
| Vaccine Type | Required Storage Temperature | Shelf Life | Field Handling |
|---|---|---|---|
| Live attenuated (freeze-dried) | 2–8°C (never frozen after reconstitution) | Per manufacturer label | Use within 1–2 hours of reconstitution; protect from sunlight and heat |
| Inactivated oil-emulsion | 2–8°C (never frozen — freezing destroys emulsion) | Per manufacturer label | Warm to room temperature before injection; shake well |
| Marek’s HVT (cell-associated) | Liquid nitrogen (−196°C) or frozen ampule (−70°C) | Per manufacturer label | Handle with cryogenic protection; use immediately on thawing |
Practical Cold Chain Protocol for Field Vaccination
- Transport vaccines to the farm in an insulated cooler with ice packs. Ice packs should be wrapped to prevent direct contact with vaccine vials — direct ice contact can freeze live vaccines and destroy cell structure.
- Do not remove vaccines from the cooler until the moment of use.
- Record vaccine batch number, manufacture date, expiry date, and storage temperature log for every vaccination event. This record is the evidence base for investigating any vaccine failure.
- Never return unused reconstituted live vaccine to the vial or store it for later use. Discard all unused reconstituted vaccine within 2 hours.
- Do not vaccinate sick or stressed birds. Immune response to vaccination in a bird already under pathogen challenge or environmental stress is significantly weaker than in a healthy bird. Postpone vaccination until the stressor is resolved.
Chlorine and Vaccine Incompatibility
Municipal water and treated borehole water commonly contain 0.3–0.5 ppm residual chlorine — sufficient to partially inactivate live vaccine virus within minutes of mixing. Before drinking water vaccination, either use unchlorinated water or add skim milk powder at 2–4 g/liter to the water 5 minutes before adding the vaccine. The protein in skim milk binds free chlorine and provides a protective colloidal matrix that extends vaccine stability in the water solution.
Verifying Vaccination Efficacy: Serology as the Feedback Loop
Vaccination is an input. Serology is the measurement of its output. A vaccination program without periodic serological monitoring is a program without feedback — you cannot know whether the schedule is producing protective immunity until a disease event proves it was not.
When to Test
- At day 1: Maternal antibody ELISA for NDV and IBD to calibrate first vaccination timing to actual maternal titer levels in the incoming flock.
- At week 4–5: Post-primary IBD serology to confirm that the Gumboro vaccination bridged the maternal antibody window and produced active seroconversion.
- At week 12: NDV HI (hemagglutination inhibition) titers to confirm that the rearing vaccination program has established adequate protective titers before the inactivated booster.
- At week 18 (pre-transfer): Full panel — NDV HI, IBD ELISA, IBV ELISA — to confirm that the complete rearing program has produced protection across all three primary pathogens before the flock enters the laying house.
Interpreting Results
HI titers for Newcastle disease of 4 log₂ or above (equivalent to ≥1:16) indicate protective immunity at the individual bird level. Flock average HI titers of 6 log₂ or above at pre-transfer indicate adequate herd-level protection. Titers below 4 log₂ in more than 20% of sampled birds at week 18 indicate vaccination program failure requiring investigation before transfer.
IBD ELISA titers that show high variation between birds (wide range from very low to very high within the same flock) indicate that vaccination did not produce uniform seroconversion — some birds were protected, and others were not. This pattern is characteristic of timing failure during the maternal antibody window: some birds had already lost maternal protection (and were protected by the vaccine), while others still had high maternal titers (and the vaccine was neutralized before it could replicate).
Common Vaccination Failures and Their Causes
Neutralization by maternal antibodies. First NDV or IBD vaccination given too early. Extend the interval; consider maternal antibody mapping before scheduling the first dose for new parent flocks.
Vaccine inactivated by chlorine in drinking water. No skim milk added before vaccination. Add 2–4 g skim milk powder per liter of vaccination water.
Cold chain break. Vaccine stored or transported above 8°C. Reconstruct the cold chain at every link and confirm temperature logs show no excursions.
Underdosing through drinking water. Birds did not drink the target volume during the vaccination period. Extend water withdrawal to ensure birds are thirsty; verify all drinkers are functional and accessible.
Wrong strain for circulating field pathogen. The Massachusetts IBV vaccine is used where 793B or a local variant is the predominant field strain. Commission field strain identification through a veterinary diagnostic laboratory and adjust vaccine strain selection.
Vaccination of immunosuppressed birds. Gumboro infection before vaccination destroyed the bursa; subsequent vaccinations produced no protective response. Test for subclinical IBD in early mortality birds. Adjust IBD vaccination timing.
Fowl pox has a rate below 90%. Incorrect wing web stab technique, compromised vaccine viability, or birds already immune. Retrain the vaccination team; replace the vaccine from a confirmed cold chain source; check for prior field exposure.
Biosecurity as Vaccination’s Complement
Vaccination reduces the probability and severity of disease challenge. It does not eliminate field pathogen exposure. A vaccination program that is executed correctly and completely, but operates in a farm with poor biosecurity, is managing a disease pressure that is continuously higher than necessary.
The two systems are complementary, and both are required. A farm with strong biosecurity and a weak vaccination program is exposed to risk from the pathogens the program missed. A farm with a strong vaccination program and weak biosecurity is fighting field challenge levels that overwhelm even correctly administered vaccines.
Vaccination protects against the pathogens you anticipate. Biosecurity reduces exposure to the pathogens you did not.
Summary
The pullet vaccination schedule from week 1 to week 18 is not a checklist. It is a phased immunological construction project — building active immunity against each priority pathogen in sequence, timed to the bird’s changing immune capacity, and verified by serology before the flock enters the laying house.
Every element of this program depends on the one before it: maternal antibody levels determine first vaccination timing; first vaccination produces the primary response that the booster amplifies; the booster produces the titer that the inactivated pre-transfer vaccine elevates to a level that protects through early lay. Remove any link, and the chain does not hold.
The flock that enters the laying house with full, verified immune protection against Newcastle disease, Gumboro, infectious bronchitis, fowl pox, and infectious coryza will produce for 52 weeks without a disease-driven production collapse. The flock that enters with gaps in that protection will produce until one of those gaps is found by a pathogen — and in West Africa, the wait is rarely long.