Neither Escherichia coli nor Salmonella requires an introduction. Both have been present in poultry houses since commercial egg production began. Both continue to cause significant mortality, production loss, and food safety liability in layer operations across West and Central Africa — not because effective control tools do not exist, but because both organisms thrive precisely where management has gaps: in wet litter, contaminated water lines, poorly cleaned equipment, overcrowded housing, and flocks immunocompromised by viral disease or nutritional deficiency.
There is a second layer to this problem that is specific to large flocks. A 500-bird backyard operation with an E. coli problem loses a manageable number of birds. A 10,000-bird commercial layer house with the same E. coli problem — where the pathogen has access to the same water line, the same feeder system, and the same shared air space as every bird simultaneously — does not scale linearly. It scales catastrophically. The management systems that prevent these pathogens from reaching critical flock prevalence in large commercial operations are different in degree, and sometimes in kind, from what is adequate in small flocks.
This article covers both pathogens with the specificity that large-flock commercial management requires: the biology of each organism, why they succeed in poultry houses, the clinical presentations that distinguish them from each other and from viral causes, the treatment decisions and their limitations, and the systems-level prevention protocols that are the only sustainable approach to control at a commercial scale.
Escherichia coli: Understanding the Opportunist
What E. coli Is — and Is Not
E. coli is a gram-negative, facultative anaerobic bacterium that is a normal component of the gastrointestinal microbiome of healthy poultry. Most E. coli strains in the poultry gut are non-pathogenic commensals that perform useful functions — competing with pathogenic bacteria for colonization sites and nutrients.
The strains that cause disease — collectively called Avian Pathogenic E. coli (APEC) — are a subset that possess specific virulence factors: type 1 and type P fimbriae for adhesion to respiratory and urinary epithelium, aerobactin siderophores for iron acquisition in host tissue, and serum resistance factors that allow survival in blood after systemic invasion. APEC strains are most commonly found in serogroups O1, O2, O35, and O78 — the same serogroups that dominate E. coli disease in commercial poultry globally.
The distinction matters because E. coli disease in poultry is almost always a secondary or opportunistic condition — not a primary pathogen that attacks immunologically competent, well-nourished birds. The clinical question is never only “E. coli or not?” It is always “E. coli on top of what?”
The Predisposing Factors That Allow APEC to Become Pathogenic
Immunocompromise: Infectious Bursal Disease destroys B-lymphocytes that produce antibodies against E. coli. Newcastle disease virus damages the respiratory mucosa, the primary barrier against E. coli invasion of the respiratory tract. Marek’s disease and chronic Mycoplasma infection impair T-cell function. Any flock that has experienced viral immunosuppression is at elevated E. coli risk regardless of the bacterial challenge level.
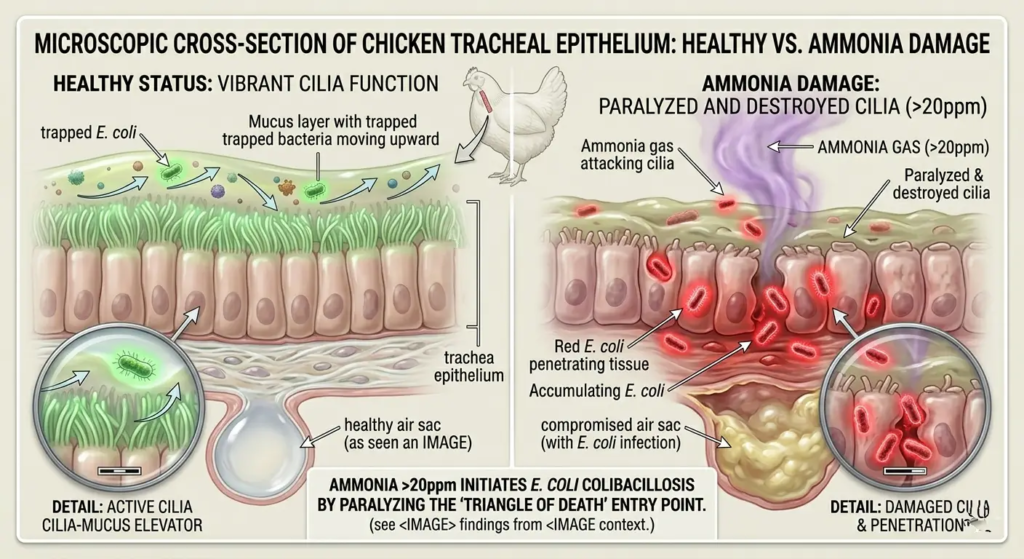
Respiratory mucosal damage: E. coli enters the respiratory tract through inhaled fecal dust. Intact mucociliary clearance — the coordinated beating of cilia in the tracheal and bronchial epithelium — normally removes inhaled bacteria before they can colonize. Ammonia above 20 ppm paralyzes the cilia. Infectious Bronchitis virus damages the epithelium. Either disruption allows APEC to penetrate and cause airsacculitis — inflammation of the air sacs — which is the most common presentation of E. coli disease in commercial layer flocks.
Water contamination: E. coli multiplying in contaminated drinker lines and bell drinkers is ingested at high concentrations throughout the day. Once in the gastrointestinal tract at high doses, even partially virulent strains can cross the intestinal barrier, particularly in birds whose gut integrity has been compromised by coccidiosis or heat stress-induced villus atrophy.
High stocking density: Fecal-oral transmission of APEC accelerates with density. In a 10,000-bird house with contaminated litter, every bird is continuously inhaling and ingesting fecal material containing APEC at concentrations that overwhelm normal immune clearance — particularly in younger, less immune-experienced flocks.
Clinical Presentations of E. coli Disease in Layers
E. coli disease in commercial layers is a diverse syndrome that changes presentation depending on the route of entry, the age of the bird, and the concurrent predisposing conditions.
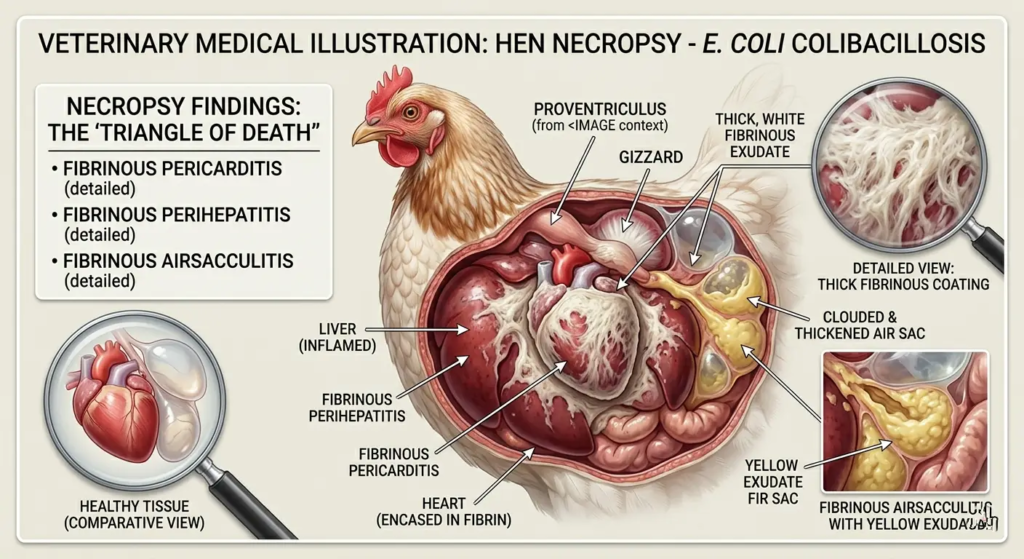
Colibacillosis (Airsacculitis / Chronic Respiratory Disease Complex): The most common presentation in laying flocks. APEC invades the respiratory tract — usually following Mycoplasma gallisepticum or IBV damage to the respiratory epithelium — and colonizes the air sacs, pericardium, and peritoneum.
Clinical signs:
- Respiratory distress — tracheal rales, labored breathing, extended neck breathing
- Reduced feed and water intake
- Progressive mortality over 1–4 weeks; not the sudden mortality spike of NDV
- Mortality rate 2–15%, depending on predisposing viral status and treatment response
Gross pathology:
- Air sac thickening and cloudy exudate (caseous or mucopurulent material) filling air sac spaces
- Pericarditis — fibrinous exudate surrounding the heart
- Perihepatitis — fibrinous exudate covering the liver surface
- “Triangle of death”: the characteristic combination of airsacculitis + pericarditis + perihepatitis at necropsy is pathognomonic for colibacillosis
Egg Peritonitis (Salpingoperitonitis / Egg Yolk Peritonitis): APEC infects the oviduct and peritoneum through retrograde contamination from the cloaca — particularly common in peak-laying hens. Yolk material from broken or mispositioned follicles provides a nutrient-rich medium for bacterial proliferation in the abdominal cavity.
Clinical signs:
- Swollen, “penguin stance” abdomen — distended from fluid and yolk accumulation
- Sudden death in otherwise apparently healthy hens at peak production — the “peak death syndrome” associated with E. coli peritonitis
- Cessation of laying in affected birds before death
- Affects individual birds rather than the whole flock simultaneously — distinguishable from a virus-driven flock-wide event
Gross pathology:
- Peritoneal cavity filled with yolk material — coagulated, caseous, and malodorous
- Oviduct congestion and hemorrhage
- Severe peritoneal inflammation
This presentation is responsible for a significant proportion of mortality in the first 10 weeks of lay in commercial layer flocks — often misattributed to “nutritional problems” or “transfer stress” rather than correctly diagnosed as E. coli salpingoperitonitis.
Septicemia: Systemic E. coli infection following any breach of mucosal barriers. Results in acute death — the bird is found dead without obvious premonitory clinical signs. At necropsy: general visceral congestion, enlarged and friable liver with or without perihepatitis, petechial hemorrhages on the heart surface.
Omphalitis (Navel Infection in Chicks): APEC enters through the incompletely closed umbilicus in day-old chicks — either from contamination in the hatchery environment or from contaminated litter in the brooding house. Affected chicks show swollen, moist, or dark navels, weakness, and death in the first 3–7 days. A hatchery problem requiring veterinary notification to the supplier.
Salmonella: The Food Safety Pathogen
Why Salmonella Is Different
Salmonella in layer flocks is a fundamentally different problem from E. coli. While E. coli is primarily an animal health and production problem, Salmonella is simultaneously an animal health problem, a public health problem, and a legal liability.
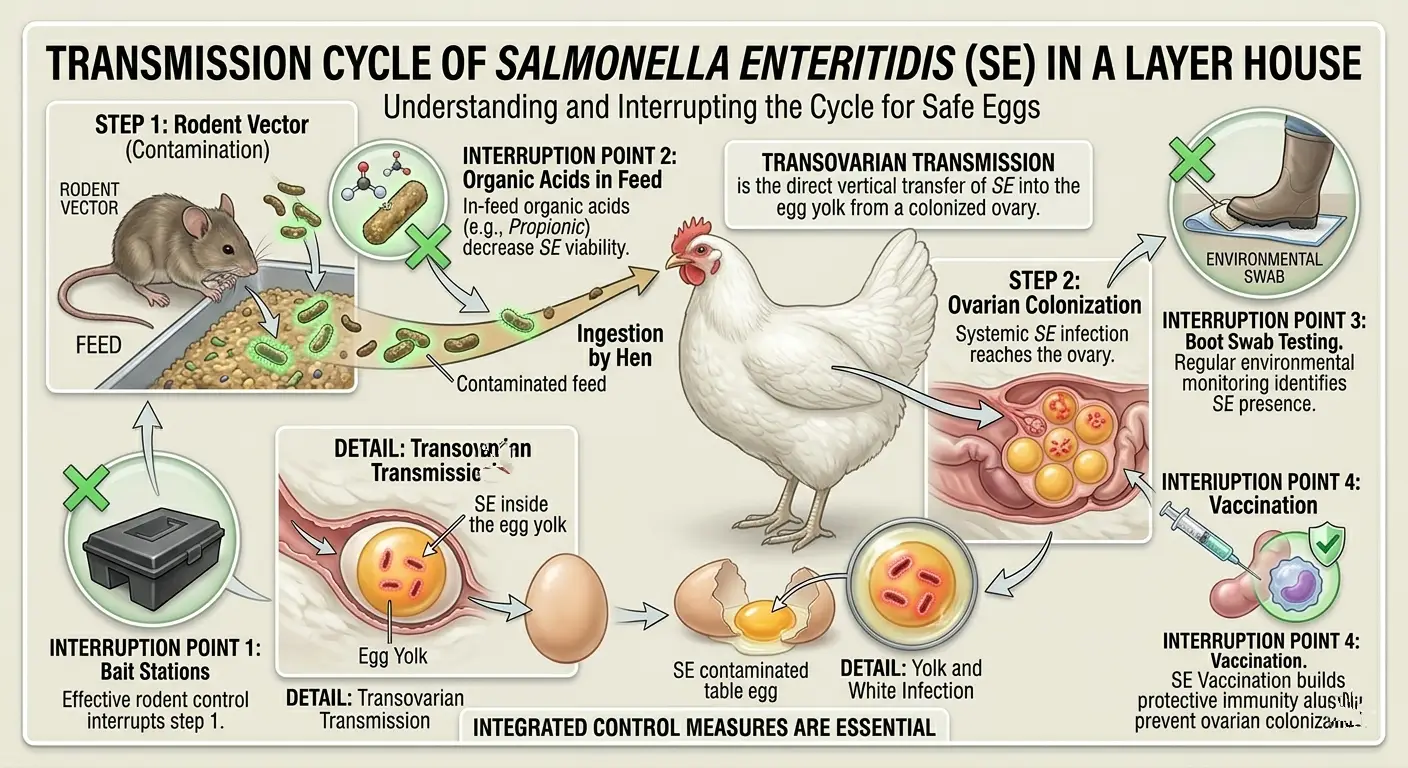
Salmonella Enteritidis (SE) and Salmonella Typhimurium (ST) — the two serovars of primary concern in egg production — can be deposited in the egg content before the shell is formed (transovarian contamination), making the laid egg a vehicle for human salmonellosis independent of shell surface contamination. A hotel or restaurant buyer who traces a salmonellosis cluster in guests to eggs from a specific farm does not simply stop buying from that farm. The consequences include food safety investigations, farm closure orders, and potential criminal liability in markets with food safety legislation.
In Cameroon, Nigeria, and Ghana, food safety regulatory frameworks are evolving. Salmonella monitoring requirements for commercial egg production are not yet uniformly enforced at the level of European or North American markets — but premium buyers (hotels, supermarkets, and export channels) increasingly require Salmonella-free certification or testing documentation as a condition of supply contracts. Managing Salmonella correctly is not only an animal health obligation — it is a market access requirement for any producer targeting above-commodity pricing.
Salmonella Biology in Laying Flocks
Salmonella Enteritidis has evolved a specific adaptation to the laying hen reproductive tract. It colonizes the ovary and upper oviduct of infected hens without causing visible clinical signs in many cases, meaning a hen can be shedding SE into eggs while appearing clinically healthy and maintaining normal production.
Transmission routes in layer operations:
- Horizontal spread: Fecal-oral transmission through contaminated water, feed, litter, and equipment between birds in the same house
- Vertical transmission: Salmonella-contaminated feed or environment leading to crop colonization in laying hens, with subsequent ovarian contamination — the route for transovarian egg content contamination
- Environmental persistence: Salmonella survives in poultry house environments for months to years in dried fecal material, litter, and dust. A house with a Salmonella history that has not been decontaminated to a verified standard will reinfect the next flock.
- Rodent vectors: Rodents are the primary external reservoir for Salmonella introduction to a previously clean flock. Rat and mouse populations in or around poultry houses are a critical Salmonella entry point that no vaccination program can compensate for.
Clinical Signs of Salmonella in Layers
Salmonella Pullorum (Pullorum Disease): Primarily a disease of young chicks — week 1–3 — causing diarrhea, huddling, depression, and mortality of 5–20%. Characteristically associated with white, pasty droppings adhering to the vent feathers. Older birds that survive become carriers. Pullorum is a notifiable disease in most countries and is the target of the Pullorum-Typhoid agglutination test used in formal flock health programs.
Salmonella Gallinarum (Fowl Typhoid): Primarily affects adult hens. Causes severe septicemia with mortality of 10–60% in susceptible flocks. Clinical signs: marked depression, anorexia, intense yellow-green diarrhea, anemia (pale combs and wattles from hemolytic anemia), and sudden death. At necropsy: enlarged, congested liver with characteristic bronze discoloration; enlarged, congested spleen; swollen kidneys. Fowl Typhoid is a serious production-level crisis event.
Salmonella Enteritidis and Typhimurium: The food safety serovars. Clinical signs in adult hens are frequently absent or non-specific — reduced production, increased mortality from secondary E. coli septicemia, and occasional diarrhea. The majority of SE-contaminated flocks do not present with a recognizable disease syndrome. Detection requires microbiological surveillance, not clinical observation.
Antibiotic Treatment: What Works, What Does Not, and Why It Matters
Treatment of E. coli Disease
E. coli disease in layers responds to antibiotic treatment — but the response depends critically on whether the antibiotic used matches the susceptibility profile of the actual APEC strain circulating in the flock.
Antibiotic selection without susceptibility testing — prescribing amoxicillin, tetracycline, or enrofloxacin based on habit rather than culture and sensitivity — produces three problems simultaneously:
- Treatment failure when the strain is resistant to the chosen antibiotic
- Selection pressure that accelerates resistance development in the farm’s bacterial population
- Potential food safety violations if the antibiotic used is in a category not approved for laying hens or if egg withdrawal periods are not observed
Recommended protocol:
- Collect fresh dead birds (within 4 hours of death) or moribund birds for necropsy and bacteriology
- Submit tissues (liver, spleen, air sac swabs, oviduct, where appropriate) to a veterinary diagnostic laboratory for bacterial culture, isolation, and sensitivity testing
- Await sensitivity results (48–72 hours) before initiating treatment, except in cases of acute high mortality, where empirical treatment must begin immediately.
- Initiate empirical treatment with the antibiotic most likely to be effective based on regional resistance patterns — in West Africa, first-line empirical choices include: amoxicillin-clavulanate (for mixed E. coli + secondary bacterial infections), doxycycline (broad-spectrum, low cost, good egg safety profile), or trimethoprim-sulfamethoxazole (good E. coli activity, avoid in laying hens due to potential effects on egg quality at high doses)
- Adjust to the sensitivity-guided antibiotic when culture results are available.
- Observe all egg withdrawal periods — most antibiotics require a 7–14 day withdrawal from eggs before the flock can return to commercial egg production.
The critical limitation of antibiotic treatment: Treatment addresses the bacterial infection. It does not address the predisposing condition that allowed E. coli to become pathogenic. A flock treated for airsacculitis that returns to the same ammonia level, the same contaminated water line, and the same unvaccinated Mycoplasma status will relapse within weeks of completing the antibiotic course.
Treatment of Salmonella Disease
Salmonella Pullorum and Gallinarum: Respond to antibiotic treatment — enrofloxacin, trimethoprim-sulfamethoxazole, or chloramphenicol (where permitted) reduce mortality. However, treatment does not eliminate the carrier state — birds that survive Pullorum or Gallinarum may remain lifelong carriers shedding Salmonella in feces without clinical signs.
Salmonella Enteritidis and Typhimurium: The food safety serovars should generally NOT be treated with antibiotics in laying flocks. Antibiotic treatment of SE in layers selects for antibiotic-resistant SE strains, which are a significantly more serious public health risk than antibiotic-sensitive strains. The correct response to confirmed SE in a laying flock is:
- Notify the relevant food safety authority (legal obligation in most jurisdictions)
- Implement enhanced flock surveillance and egg testing
- Consider the appropriate management response (depopulation, enhanced biosecurity, controlled sale) in consultation with veterinary and regulatory authorities
Antibiotic treatment of SE in laying flocks without regulatory guidance is a serious food safety error that can result in regulatory penalties and market exclusion.
The Salmonella-Free Egg Program: What It Requires
For layer producers targeting premium markets, hotel supply chains, or export channels, a Salmonella-free egg program is a market access requirement, not a voluntary quality initiative. The program has four components:
1. Flock Surveillance Testing
Environmental swabs (boot swabs, drag swabs, dust samples) from the poultry house provide Salmonella surveillance at the population level — they detect Salmonella shedding in the flock environment without requiring individual bird testing.
Protocol:
- Environmental swab 4 weeks after flock placement (confirms clean status at the start of production)
- Environmental swab every 15 weeks during lay (ongoing surveillance)
- Environmental swab before any eggs are sold to a buyer requiring Salmonella-free certification
- Blood serology (ELISA for SE antibodies) as a supplementary test to detect flocks with humoral response to SE infection
2. Rodent Control
Rodent control is the single most important preventive measure for Salmonella in commercial layer production. Rats and mice are the primary external reservoir — they carry Salmonella from contaminated external environments (neighboring farms, feed stores, waste disposal areas) into the clean flock environment, contaminate feed and water, and create fecal deposits in litter that birds contact continuously.
Rodent control program requirements:
- Continuous bait station program — not reactive baiting after rodent activity is observed. Stations positioned inside and outside the house perimeter are checked and restocked on a weekly schedule.
- Rodent entry exclusion — all wall-floor junctions sealed, mesh over ventilation openings, doors self-closing. A commercial layer house should have no gap larger than 6 mm in its perimeter — the minimum entry point size for a juvenile mouse
- Rodent population monitoring — track bait consumption as a rodent activity index. Rising consumption indicates an increasing population; falling consumption after initial baiting indicates effective control
3. Feed Safety
Salmonella-contaminated feed is a direct route to flock contamination. Commercially milled feeds from suppliers with HACCP-certified production are significantly less likely to be contaminated than on-farm mixed feeds using local ingredients.
For on-farm mixing, heat treatment of feed ingredients (pelleting at above 80°C or steam conditioning) reduces Salmonella in feed ingredients. Adding organic acids (formic acid, propionic acid) to the feed at 0.3–0.5% reduces pH in the feed matrix and inhibits Salmonella survival during storage.
4. House Decontamination Between Flocks
Salmonella is significantly more environmentally persistent than most poultry pathogens. Dried fecal material in litter, wall cracks, and cage structures can harbor viable Salmonella for 12–24 months at ambient temperature.
The between-flock decontamination protocol for Salmonella control requires:
- Complete litter removal and disposal (incinerate or compost off-site — do not spread on adjacent land that birds can access)
- Dry cleaning of all surfaces
- High-pressure hot water wash (above 60°C where possible)
- Disinfection with formaldehyde (formalin fumigation), glutaraldehyde, or a commercial quaternary ammonium compound with documented Salmonella efficacy
- Environmental swab before restocking to verify decontamination — a positive swab before restocking means the decontamination must be repeated before introducing a new flock.
Prevention System: Addressing the Root Causes in Large Flocks
Treatment is a response to failure. Prevention is the system that makes failure unlikely. In a 10,000-bird layer operation, the prevention system for both E. coli and Salmonella has seven non-negotiable components:
1. Viral Disease Vaccination and Monitoring
The most reliable way to reduce E. coli colibacillosis in a large flock is to ensure that the respiratory mucosa is not being damaged by concurrent IBV, NDV, or Mycoplasma infection. This means a correctly timed vaccination program (as covered in the vaccination article in this series), followed by serology to verify that protective immunity has been established.
Flocks that are behind on vaccination or show inadequate post-vaccination HI titers should be revaccinated before E. coli pressure increases — not treated with antibiotics after colibacillosis has already been established.
2. Ammonia Control Below 20 ppm
Ammonia above 20 ppm at the bird level paralyzes the mucociliary escalator — the primary innate immune barrier against respiratory E. coli invasion. Maintaining ammonia below this threshold is a biosecurity measure, not a welfare preference.
Ammonia is controlled by ventilation (adequate air exchange at litter level) and litter management (preventing litter moisture accumulation above 30%). Water line leaks, overfilled bell drinkers, and high-moisture feed ingredients are the most common causes of wet litter and elevated ammonia in large commercial houses.
Monitor ammonia with a portable ammonia meter (Draeger tube or electronic sensor) at bird level at three points in the house monthly. If any point exceeds 20 ppm during production hours, investigate the moisture source before the next flock health check.
3. Water Line Sanitation
As covered in the water quality article in this series, E. coli multiplying in biofilm-coated water lines is the highest-concentration E. coli challenge most flocks experience. Waterline sanitization with 3–5 ppm free residual chlorine maintained at the far-end nipple during production — and shock chlorination with hydrogen peroxide or peroxygen compounds between flocks — is a non-negotiable large-flock management practice.
Test the water bacterial count quarterly at the far-end nipple. A count above 100 CFU/mL at the point of consumption indicates active contamination in the distribution system, not just the source.
4. Litter Management
Dry litter is hostile to both E. coli and Salmonella. Wet litter — moisture above 30% — supports E. coli multiplication at exponential rates and maintains Salmonella viability for months.
In large floor-system or aviary houses, turn litter mechanically where possible to expose and dry wet patches. Add litter amendments (aluminum sulfate at 0.5 kg/m², or ferrous sulfate at 0.25 kg/m²) to acidify wet areas and reduce bacterial viability. Do not cover wet patches with fresh litter — the moisture is still present, and the problem continues underneath the dry top layer.
5. Stocking Density Management
Fecal-oral transmission of both pathogens is density-dependent. At the stocking densities appropriate for the ventilation system (25–30 kg/m² for natural ventilation, 35–40 kg/m² for tunnel ventilation), the fecal challenge per bird is managed. Above these densities, it is not.
Do not overstock. The marginal revenue from additional birds per square meter is almost always outweighed by the increased disease pressure, increased mortality, and increased treatment cost that above-threshold density produces.
6. Probiotic and Organic Acid Supplementation
Competitive exclusion — populating the gastrointestinal tract with commensal bacteria that compete with APEC and Salmonella for colonization sites — is the most evidence-based non-antibiotic intervention for E. coli and Salmonella control.
Probiotics: Multi-strain preparations containing Lactobacillus, Bacillus subtilis, Enterococcus faecium, and Saccharomyces cerevisiae have documented competitive exclusion efficacy against both APEC and Salmonella in commercial poultry. Administer on the day of hatch (in drinking water) and continuously through the rearing and laying periods at the label dose. In flocks with a history of Salmonella or E. coli problems, probiotic supplementation is one of the most cost-effective preventive investments available.
Organic acids: Formic acid, propionic acid, butyric acid, and lactic acid — individually or in commercial blended acid products — reduce gut pH to levels that inhibit APEC and Salmonella colonization. Particularly effective when combined with probiotic supplementation: the acid creates the hostile environment; the probiotics populate it. Target pH in the crop and proventriculus below 4.0.
7. Dead Bird Management
Dead birds in a commercial layer house are point-source concentrations of every pathogen active in the flock. A bird dead from colibacillosis is shedding APEC at fecal concentrations. A dead bird from Fowl Typhoid is a Salmonella reservoir being approached and potentially pecked by living flock-mates.
Collect dead birds twice daily in large flocks (once daily is inadequate in a 10,000-bird house). Store in sealed, labeled bags in a dedicated dead bird refrigerator or secure covered container. Dispose of through rendering, incineration, or composting — not open dumping, which spreads pathogens to wild birds and rodents that re-enter the farm environment.
The Investigation Sequence: When E. coli or Salmonella Is Suspected
- Collect fresh dead birds or moribund birds — within 4 hours of death for bacteriology. Submit to a veterinary diagnostic laboratory with a completed submission form specifying suspected diagnosis and requested tests (culture, sensitivity, Salmonella serotyping if applicable).
- Take water samples at the far-end nipple and submit for bacterial culture simultaneously. If the water is contaminated, the treatment is not working correctly.
- Measure ammonia at the bird level at three points in the house. Record the readings. If above 20 ppm, investigate the moisture source before adjusting the treatment plan.
- Review the vaccination record for the last 90 days. Was NDV or IBV revaccination due and missed? Was the Gumboro vaccination correctly timed? Immune suppression from viral disease is the most common predisposing factor for E. coli colibacillosis.
- Await sensitivity results before finalizing antibiotic selection. Initiate empirical treatment if mortality exceeds 0.2% per day while awaiting results.
- Notify the relevant food safety authority if Salmonella Enteritidis or Typhimurium is confirmed. This is a legal obligation, not an option.
Summary
E. coli and Salmonella are bacterial pathogens that succeed in layer operations where management creates the conditions for their success — wet litter, contaminated water, immunosuppressed birds, ammonia-damaged respiratory mucosa, and rodent access. Neither is an unstoppable force. Both are predictable opportunists.
The control program for a large layer flock is a systems response, not a treatment response: viral immunity maintained through correctly executed vaccination, ammonia controlled through ventilation and litter management, water lines kept below 100 CFU/mL through active sanitization, rodent populations kept out through exclusion and continuous baiting, and competitive exclusion maintained through probiotic and organic acid supplementation.
Antibiotic treatment is the response to a system that has already failed. The goal is a management system that fails infrequently enough that antibiotic treatment is a rare event, not the primary production management tool that it becomes when everything upstream is not working.
The farms that manage E. coli and Salmonella well do not treat them less effectively. They create environments where these pathogens succeed less frequently.