Parasites do not announce themselves with mortality spikes or respiratory distress. They take production slowly — a gram of egg weight here, a percentage point of laying rate there, a unit of FCR every week — in ways that accumulate invisibly until a flock that should be producing 88% is producing 74% and nobody can explain why without looking closely at what is living on and inside the birds.
In commercial layer operations across West and Central Africa, two parasite categories consistently cause undiagnosed production losses: external parasites — principally the red poultry mite (Dermanyssus gallinae) and the northern fowl mite (Ornithonyssus sylviarum) — and internal parasites, primarily the roundworm Ascaridia galli, the cecal worm Heterakis gallinarum, and the tapeworm species Raillietina spp.
Both categories are more prevalent than most flock health monitoring programs detect. Both are more economically damaging than their clinical presentations suggest. And both are manageable through a combination of accurate detection, correctly timed treatment, and environmental control that removes the conditions that allow populations to establish and reestablish between production cycles.
This article covers the biology, detection methods, clinical signs, treatment protocols, and environmental management for both parasite categories, with specific attention to the conditions that make layer flocks in tropical West Africa particularly vulnerable.
Part 1: External Parasites
The Red Poultry Mite (Dermanyssus gallinae)
Biology and Life Cycle
Dermanyssus gallinae is the most economically damaging external parasite of laying hens globally. It is an obligate hematophagous ectoparasite — meaning it feeds exclusively on blood — but unlike most bloodsucking parasites, it does not live on the bird. It lives in the house infrastructure: in cracks in cage frames, wall junctions, perch surfaces, nest box corners, and any dark, sheltered microhabitat within the house where it can aggregate and reproduce.
The life cycle from egg to reproductive adult takes 7–10 days at optimal temperature (25–30°C) — exactly the conditions in a tropical layer house — and 5–10 days at reproductive temperatures (20–35°C). A single female mite lays 6–8 eggs per blood meal. A population can double every 1–2 weeks under optimal conditions.
The mite feeds on birds at night — moving from its harborage in the house structure to feed on resting hens during the dark period (typically 1–2 hours after lights-out), then returning to the structure before lights-on. During the day, there are no mites on the birds, but the house structure is densely infested with all life stages. A farm worker inspecting birds in daylight will find no mites on the birds and incorrectly conclude that mites are not present.
This nocturnal feeding pattern is the primary reason red poultry mite is chronically underdiagnosed in commercial layer operations.
Detection Methods
The white cloth test (definitive field test): After lights-out, wait 1–2 hours, then enter the house with a flashlight. Run a piece of white cloth or white paper along the underside of perches, along cage frame joints, into nest box corners, and into any visible cracks in the structure. If mites are present, they will be visible on the white surface as tiny moving red or dark red dots (engorged with blood) or pale-colored moving dots (unfed). Moving dots smaller than 1 mm in size are diagnostic for Dermanyssus.
The sticky trap method: Place double-sided sticky tape wrapped around wooden dowels or cardboard tubes into suspected harborage sites — perch ends, cage frame corners — before lights-out and remove them 2 hours after lights-out. Count the mites captured per trap. This provides an infestation intensity index that can be used to track population trends over time and to evaluate treatment efficacy.
Bird examination at night: Two hours after lights-out, examine the vent area, under the wings, around the head, and on the breast of randomly selected birds using a flashlight and magnifying glass. Mites actively feeding on birds will be visible as moving dark red or pale specks. Heavy infestations make the feathers appear as if they are “moving” under direct examination.
Daytime indicators (indirect detection):
- Restless behavior at night — hens disturbed by feeding mites may show increased nighttime activity visible in mortality records or through CCTV if installed
- Anemia signs: pale combs and wattles, pale breast muscle at necropsy — from chronic blood loss in heavy infestations
- Reduced laying rate and egg weight without other identifiable cause during an otherwise stable period
- Skin irritation signs — hens with heavy mite burden show increased preening frequency, feather damage, and self-inflicted skin excoriation
The Production Impact of Red Poultry Mite
The economic damage from Dermanyssus is dose-dependent — meaning it scales with infestation intensity. Light infestations (detected but below population threshold) cause measurable but modest production losses. Heavy infestations cause severe losses.
Quantified production effects from controlled research:
| Infestation Level | Effect on Laying Rate | Effect on Feed Intake | Estimated Egg Production Loss |
|---|---|---|---|
| Moderate (500–2,000 mites/trap/night) | −3–6% | +5–8% | 8–15 eggs/hen/year |
| Heavy (2,000–10,000 mites/trap/night) | −8–15% | +10–15% | 20–40 eggs/hen/year |
| Severe (>10,000 mites/trap/night) | −15–30% | +15–25% | 40–80 eggs/hen/year + mortality |
Feed intake increases simultaneously with production loss because mite-infested birds are in a chronic stress state that elevates corticosterone and metabolic rate — they consume more feed to compensate for the energy cost of immune activation against blood-sucking and the thermal response to stress, while simultaneously producing fewer eggs.
At heavy infestation levels in a 5,000-bird flock, the production loss exceeds 150,000 eggs per laying cycle. At prevailing commercial egg prices, this represents revenue loss that dwarfs the cost of any mite treatment program many times over.
Treatment Protocol for Red Poultry Mite
Critical principle: Treating the birds with an acaricide (mite-killing agent) while leaving the house structure untreated produces temporary mite count reduction on birds but leaves the environmental population intact. Within 2–4 days, mites from the structure reinvade, and the population recovers fully. Effective treatment must target the house structure — the primary habitat — with sufficient thoroughness to eliminate all life stages, including eggs.
Step 1 — House treatment (primary intervention):
Apply an approved acaricide to all surfaces in the poultry house — cage frames, perches, nest boxes, wall-floor junctions, ventilation ducts, electrical conduit surfaces, and any identified harborage sites — using a pressure sprayer that achieves penetration into cracks and joints.
Acaricide options:
- Synthetic pyrethroids (cypermethrin, deltamethrin): Effective contact acaricides. Apply at the label concentration for poultry house use. Resistance is developing in Dermanyssus populations in regions with high pyrethroid use — monitor for treatment failure and rotate chemical classes.
- Organophosphates (pirimiphos-methyl): Effective against mites, longer residual activity. Use with appropriate personal protective equipment. Do not apply directly to birds or egg-contact surfaces.
- Spinosad: A fermentation product of Saccharopolyspora spinosa with good mite activity and a more favorable resistance profile than pyrethroids. Available in some markets as a commercial acaricide formulation.
- Diatomaceous earth (DE): Silica-based physical acaricide that kills mites through desiccation of their cuticle rather than chemical toxicity. No resistance development. Apply as a dust to harborage areas and perch surfaces. Less effective in high-humidity environments where the moisture reduces its desiccating action.
Step 2 — Repeat treatment at day 7–10:
The first treatment kills adults and nymphs but does not reliably penetrate egg cases. Eggs hatch 2–3 days after treatment; the next generation reaches the harborage stage by day 7–10. A second treatment within this window eliminates the emerging generation before it reproduces.
Step 3 — Between-flock elimination:
Between production cycles, apply a 2-phase treatment: acaricide application immediately after flock depopulation and litter removal, followed by a second application 10–14 days later. Allow a minimum 2-week rest period after the second treatment before restocking. During the rest period, the environmental population continues to decline without a blood meal host.
Step 4 — Ongoing monitoring:
Continue sticky trap monitoring monthly throughout the laying cycle. Populations that exceed the moderate threshold (500 mites per trap per night) require treatment. Waiting until the heavy threshold is reached before treating means treating after significant production losses have already occurred.
The Northern Fowl Mite (Ornithonyssus sylviarum)
Unlike Dermanyssus gallinae, the northern fowl mite lives permanently on the bird — it does not retreat to the house structure. The entire life cycle (egg to egg in 5–7 days) occurs on the bird’s body, primarily around the vent, under the wings, and on the breast.
Detection: Examine the vent area and under the wings of 20–30 randomly selected birds in daylight. Northern fowl mites are visible as tiny dark specks moving in the feather tracts. The vent feathers of heavily infested birds appear darkened and crusted from the accumulation of mite feces, egg shells, and shed mite skin.
Treatment: Because the mite lives on the bird, on-bird treatment with an approved pyrethroid spray or dust is effective and directly targets the population. Treat the entire flock simultaneously — treating individual birds allows the untreated population to reinvade treated birds within days.
Part 2: Internal Parasites
Ascaridia galli (Large Intestinal Roundworm)
Biology and Life Cycle
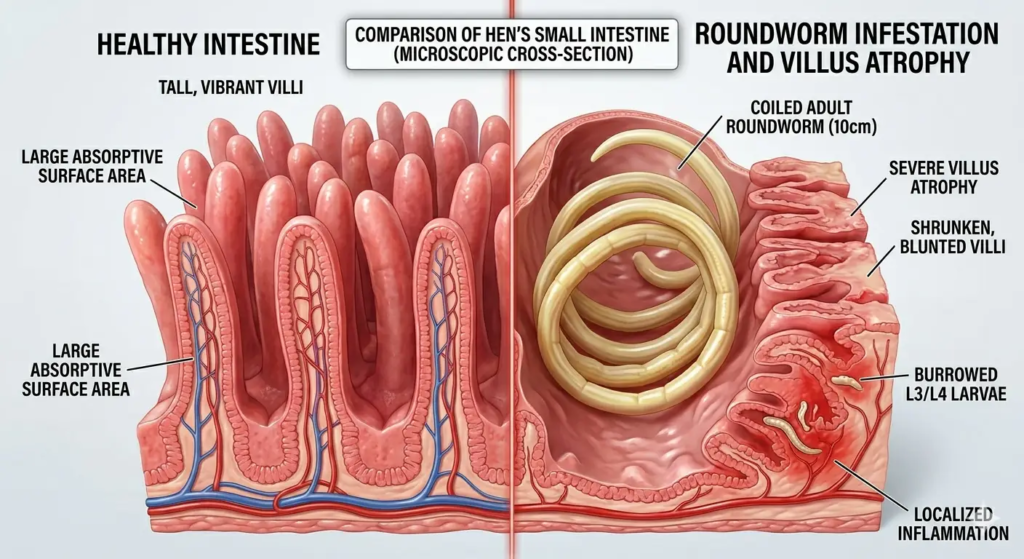
Ascaridia galli is the most prevalent intestinal helminth in layer flocks managed on litter or in aviary systems. It is a large nematode — adults reach 5–11 cm in length — visible to the naked eye in the small intestine at necropsy.
The life cycle is direct (no intermediate host required): eggs passed in feces embryonate in the litter environment in 10–12 days at optimal temperature and humidity, becoming infective L2 larvae. Hens ingest infective eggs from contaminated litter, water, or feed. Larvae penetrate the intestinal epithelium, develop through the L3 and L4 stages (during which they are embedded in the intestinal wall and cause the most tissue damage), and emerge as L5 adults that live in the lumen of the small intestine. The full prepatent period (from ingestion to egg production) is 35–60 days.
The embedded larval stages cause the most significant production impact — intestinal inflammation, villus atrophy, and reduced absorptive capacity — during a 3–6-week window that is invisible without necropsy.
Clinical Signs and Production Impact
Light worm burdens (below 50 adult worms per bird) cause subclinical production losses — reduced FCR and egg weight — without clear visible signs. These light burdens are the ones most commonly missed because they produce no mortality and no acute clinical presentation.
Heavy burdens (above 200 adult worms) produce:
- Reduced feed intake and loss of body condition
- Diarrhea — watery droppings with abnormal color
- Pale combs and wattles from anemia
- Reduced laying rate — 10–20% below expected trajectory
- Increased mortality from intestinal obstruction in severe infestations (adult worms can physically obstruct the small intestine)
The production impact that matters economically: A flock with an average of 30–50 adult Ascaridia worms per bird throughout a 52-week laying cycle loses approximately 8–12 eggs per hen over the cycle from reduced intestinal absorption efficiency — a subclinical loss that never triggers investigation because no bird appears sick.
Detection Methods
Fecal egg count (FEC): Collect fresh fecal samples from 20–30 birds across the flock (composite sample from different house locations). Process by the McMaster flotation method using a saturated salt or sugar solution. Count nematode eggs per gram (EPG) of feces under the microscope.
Ascaridia galli eggs are large (75–90 μm), oval, with a thick outer shell and unsegmented inner content — distinctive under 10× magnification.
Interpretation:
- 0–100 EPG: Light burden; subclinical; monitor, consider strategic treatment
- 100–500 EPG: Moderate burden; significant production impact; treat
- Above 500 EPG: Heavy burden; treat immediately; investigate management factors contributing to high reinfection rate
Necropsy: Open the small intestine of freshly dead birds (within 4 hours of death) and examine the lumen for adult worms. In flocks with moderate to heavy burdens, worms are visible to the naked eye and may be present in large numbers. This is the most rapid diagnostic method when necropsy access is available.
Heterakis gallinarum (Cecal Worm)
Heterakis gallinarum is a smaller nematode (1–1.5 cm) that inhabits the ceca. It is the carrier of Histomonas meleagridis — the protozoan parasite that causes blackhead disease (histomoniasis) in turkeys and, less commonly, in chickens.
In chickens, direct Heterakis infection produces relatively mild cecal inflammation. Its significance is primarily as a vector for histomoniasis, where turkeys and chickens are kept in proximity, and as a contributor to cecal dysfunction that reduces fermentation and mineral absorption efficiency.
Detection: Same FEC method as Ascaridia — Heterakis eggs are similar in size (65–80 μm) but have more pointed poles than Ascaridia. Differentiation requires careful microscopic examination.
Tapeworms (Raillietina spp., Davainea spp.)
Tapeworms require intermediate hosts — beetles, earthworms, ants, slugs, or flies — that carry the larval cysticercoid stage. Layer hens on litter floors or with outdoor access encounter these intermediate hosts during foraging and become infected by ingesting them.
Clinical Impact
Adult tapeworms attach to the intestinal mucosa via scolex hooks and suckers, causing local mucosal damage, bleeding, and inflammatory infiltration at attachment sites. Heavy tapeworm burdens produce:
- Diarrhea with mucus — from intestinal mucosal irritation
- Progressive weight loss and reduced body condition
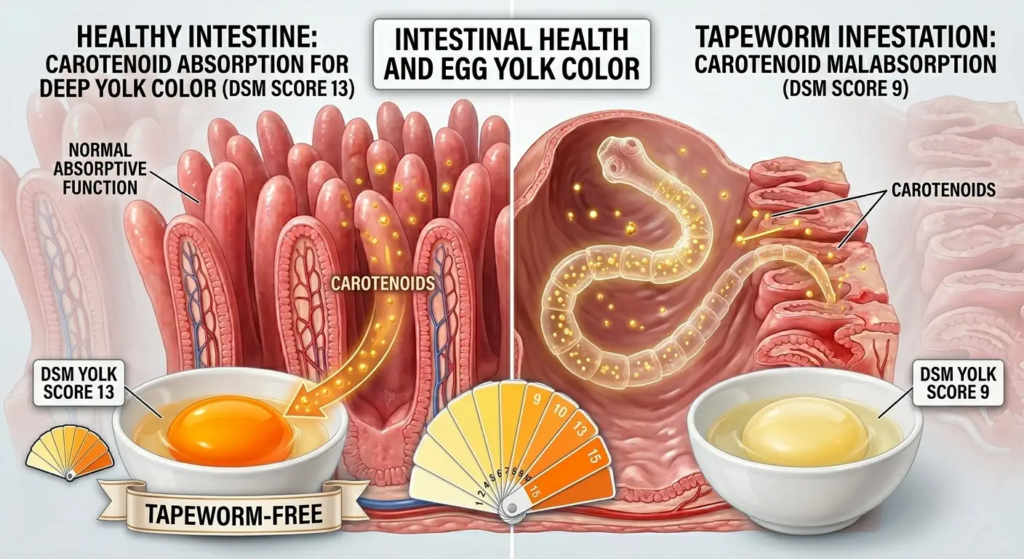
- Pale yolks (carotenoid competition between parasite and host — tapeworms accumulate carotenoids preferentially, reducing yolk pigmentation)
- Reduced laying rate in heavy infestations
The pale yolk effect — detectable by monitoring DSM Yolk Color Fan scores — is a useful indirect indicator of tapeworm burden in flocks on pigmented rations where yolk color should be stable.
Detection: FEC for tapeworm proglottids (segments containing eggs) is less reliable than for nematodes — proglottids are shed intermittently. Necropsy of the intestinal tract is the most reliable detection method. Open the small intestine under water — tapeworm segments detach from the mucosa and are visible floating in the wash water.
Integrated Parasite Management: The Framework
Treating parasites without addressing the conditions that allow them to establish and reestablish between treatment cycles produces a treatment program that never ends and costs more than prevention.
Integrated parasite management combines anthelmintic treatment with environmental management and monitoring to reduce transmission risk alongside pathogen load.
Anthelmintic Treatment
For Ascaridia galli and Heterakis gallinarum:
| Drug | Dose | Route | Efficacy Against | Egg Withdrawal |
|---|---|---|---|---|
| Fenbendazole | 5–10 mg/kg body weight × 5 days | Drinking water or feed | Ascaridia (all stages), Heterakis | 7 days |
| Flubendazole | 1 mg/kg × 7 days | In feed | Ascaridia, Heterakis | 6 days |
| Levamisole | 30–40 mg/kg (single dose) | Drinking water | Ascaridia adults (not larvae) | 3 days |
| Piperazine | 100–200 mg/kg × 2 days | Drinking water | Ascaridia adults only | 24 hours |
Critical selection guidance:
- Fenbendazole is the preferred choice because it kills larvae embedded in the intestinal wall (the L3–L4 stages responsible for the most tissue damage) as well as adult worms. Piperazine and levamisole kill only adult luminal worms — they do not address the embedded larval stages and miss the period of maximum intestinal damage.
- Repeat fenbendazole treatment at day 21–28 post-treatment to eliminate worms that were larvae at the time of first treatment and have since developed to adults.
For tapeworms (Raillietina spp., Davainea spp.):
Praziquantel (10–20 mg/kg, single dose) is the most effective cestocide (tapeworm-killing agent). It is not always available through regular veterinary channels in West Africa — source from a veterinary pharmacy or import from a regional supplier where needed.
Fenbendazole at standard doses has limited efficacy against tapeworms; do not rely on a combined fenbendazole course to address confirmed tapeworm burden.
Treatment Timing and Strategic Deworming
In layer operations on litter or aviary systems, the reinfection pressure from contaminated litter is continuous. A single deworming event followed by no further monitoring or treatment produces a temporary reduction in worm burden, followed by complete population recovery within 6–8 weeks of re-exposure to contaminated litter.
Strategic deworming program for litter-floor and aviary layer operations:
- At transfer to the laying house (week 17–18): Treat with fenbendazole before birds enter the laying house to eliminate worms acquired during rearing. This starts the laying cycle with a low worm burden.
- FEC monitoring at week 24 and week 40: If EPG exceeds 100, re-treat. If EPG remains below 100, monitor at the next scheduled point.
- At depopulation: Treat the final week before depopulation is not productive — the treatment investment has no subsequent production benefit. Instead, focus on environmental decontamination to reduce infective egg load for the next cycle.
For cage-system layer operations: Ascaridia and Heterakis transmission is significantly reduced (but not eliminated) in cage systems because hens do not contact fecal material on the litter directly. Routine strategic deworming is less critical in fully slatted or wire-floor cage systems. However, if cage-to-cage fecal contamination is occurring through water line overflow or manure belt malfunction, monitor FEC annually to verify the assumption that worm burden is low.
Environmental Management for Parasite Reduction
Litter management: Ascaridia eggs can survive in moist litter for up to 12 months at ambient temperature. Dry litter below 20–25% moisture inactivates eggs more rapidly than wet litter. Maintaining litter moisture below 30% through adequate ventilation is the most important single environmental intervention for reducing Ascaridia transmission.
Between-flock litter management: Complete litter removal between flocks eliminates the infective egg bank in the litter environment. Composting or heat-treating removes litter (internal composting temperatures above 55°C inactivate Ascaridia eggs), preventing recontamination from land application of fresh litter on adjacent land that the birds can access.
Intermediate host control for tapeworms: Reduce the beetle, earthworm, ant, and fly populations in and around the house. The primary practical interventions are: reducing litter moisture (which reduces beetle and earthworm populations), controlling fly populations through insecticide application and manure management, and preventing outdoor access where soil-dwelling intermediate hosts are the primary tapeworm source.
Rodent control: Rodents transport infective Ascaridia eggs from contaminated to clean areas of the house. Rodent control — as discussed in the E. coli and Salmonella article — is a parasite management measure as well as a bacterial disease measure.
The Monitoring Program: Seeing What Is There
The farms that manage parasites effectively are the farms that look for them — systematically and on a schedule — rather than waiting for the clinical signal that by the time it appears has already cost significant production.
Monthly External Parasite Monitoring
- Sticky trap deployment in 5 harborage sites per house, monitored monthly
- White cloth test conducted 2 hours after lights-out, monthly
- Bird examination for northern fowl mite at weekly health walk-through
Set a threshold: more than 100 mites on the white cloth test (30-second swipe) or trap counts above 200 mites per trap per night trigger a treatment response. Do not wait for trap counts above 2,000 — that threshold represents a heavy infestation that has already caused measurable production loss.
Quarterly Internal Parasite Monitoring
- FEC from composite fecal samples (20 birds minimum) every quarter in litter-based systems
- Necropsy intestinal examination on every dead bird that does not have a clear alternative cause of death
- Track EPG trends — a rising EPG trend across two consecutive quarters indicates increasing transmission pressure that requires both treatment and environmental management response.
DSM Yolk Color Fan Monitoring as a Tapeworm Indicator
In flocks on a stable pigmentation program (consistent marigold meal and paprika inclusion, no ration changes), a declining DSM score over 3–4 consecutive weeks without ration change should trigger a tapeworm investigation. Tapeworms accumulate dietary carotenoids in their tissue at the expense of yolk deposition — a flock with moderate tapeworm burden may show a 1–3 unit DSM decline that precedes other clinical signs by 4–6 weeks.
The Economic Case for Active Parasite Management
The production losses from uncontrolled parasite burden in a 5,000-bird layer operation:
- Red poultry mite at heavy infestation: 40–80 eggs lost per hen per year × 5,000 hens = 200,000–400,000 eggs per cycle
- Ascaridia at moderate burden (30–50 worms/bird): 8–12 eggs lost per hen per cycle × 5,000 hens = 40,000–60,000 eggs per cycle
- Tapeworm (moderate burden, 3–4 DSM unit yolk color reduction): Premium market downgrade or rejection on color specification — loss of price premium on affected production
Combined, uncontrolled parasitism in a moderately infested flock represents a production shortfall equivalent to 240,000–460,000 eggs per cycle, before accounting for increased feed cost from elevated feed intake under mite stress, reduced egg quality premiums, and increased mortality.
The cost of a comprehensive parasite management program — mite monitoring and treatment, quarterly FEC, strategic fenbendazole treatment twice per cycle — is less than 1% of this potential loss.
Summary
External and internal parasites are silent production thieves. They do not announce themselves with acute mortality or diagnostic-quality clinical signs. They reduce production by degrees — laying rate, egg weight, yolk color, FCR — in ways that accumulate month by month without triggering investigation until the cumulative loss is too large to ignore.
The detection methods — white cloth test for mites, FEC for nematodes, yolk color monitoring for tapeworms, necropsy for all categories — are accessible, inexpensive, and require no specialized laboratory equipment for initial screening. What they require is the discipline to use them on a regular schedule and the knowledge to interpret what they reveal.
Parasites that are detected early are treated cheaply. Parasites that are detected after months of uncontrolled infestation are treated expensively, and the production that was lost during those months is not recovered.
Look for them. Regularly. Before the eggs stop explaining themselves.