Newcastle disease kills faster than almost anything else in commercial poultry production. A velogenic Newcastle disease virus entering an unprotected or inadequately vaccinated layer house can kill 80–100% of the flock within 7–10 days. The speed is not the worst part. The worst part is that the virus is airborne, spreads between farms on the wind and on the boots of visitors, and is endemic at such high prevalence in the backyard and village poultry population of West and Central Africa that no commercial layer farm — however well-managed — operates in a zero-exposure environment.
Gumboro disease (Infectious Bursal Disease, IBD) kills more slowly and less visibly. It does not always produce flock-wide acute mortality. What it does — reliably, silently, and at high cost — is destroy the bursa of Fabricius in young birds before their immune system has finished developing. A flock that survives a Gumboro outbreak at week 2–4 of rearing goes to the laying house immunocompromised. Every subsequent vaccination produces a weaker response than it should. Every subsequent pathogen challenge finds a weaker defense than it should. The Gumboro damage manifests as lifetime immune suppression, and the economic consequence is carried through the entire 52-week laying cycle.
Both diseases are vaccine-preventable. Both continue to cause outbreaks across West and Central Africa at a significant frequency. The gap between “vaccine-preventable” and “prevented” is almost always one of three things: a vaccination program that was not correctly designed for the actual maternal antibody status of the incoming flock, a vaccine that lost efficacy before it reached the bird due to cold chain failure, or a biosecurity environment so compromised that vaccine-induced immunity is overwhelmed by field challenge.
This article covers all three gaps — and the specific interventions that close them.
Understanding Newcastle Disease: What You Are Protecting Against
The Pathogen and Its Variants
Newcastle disease is caused by Avian Paramyxovirus type 1 (APMV-1), classified by pathogenicity into three broad groups:
Lentogenic strains (low virulence): Used as live vaccines — La Sota, Clone 30, B1. They cause mild or no clinical signs in fully susceptible birds and produce protective mucosal and systemic immunity without causing disease.
Mesogenic strains (intermediate virulence): Cause moderate respiratory and nervous system signs with variable mortality. Used as vaccines in some programs (Mukteswar, Komarov), but are not approved in many countries due to the risk.
Velogenic strains (high virulence): The field strains responsible for commercial outbreaks in West and Central Africa. Two subtypes:
- Velogenic viscerotropic (VVND): Primarily attacks the gastrointestinal tract; causes hemorrhagic lesions in the intestine and proventriculus; mortality 80–100% in unprotected flocks.
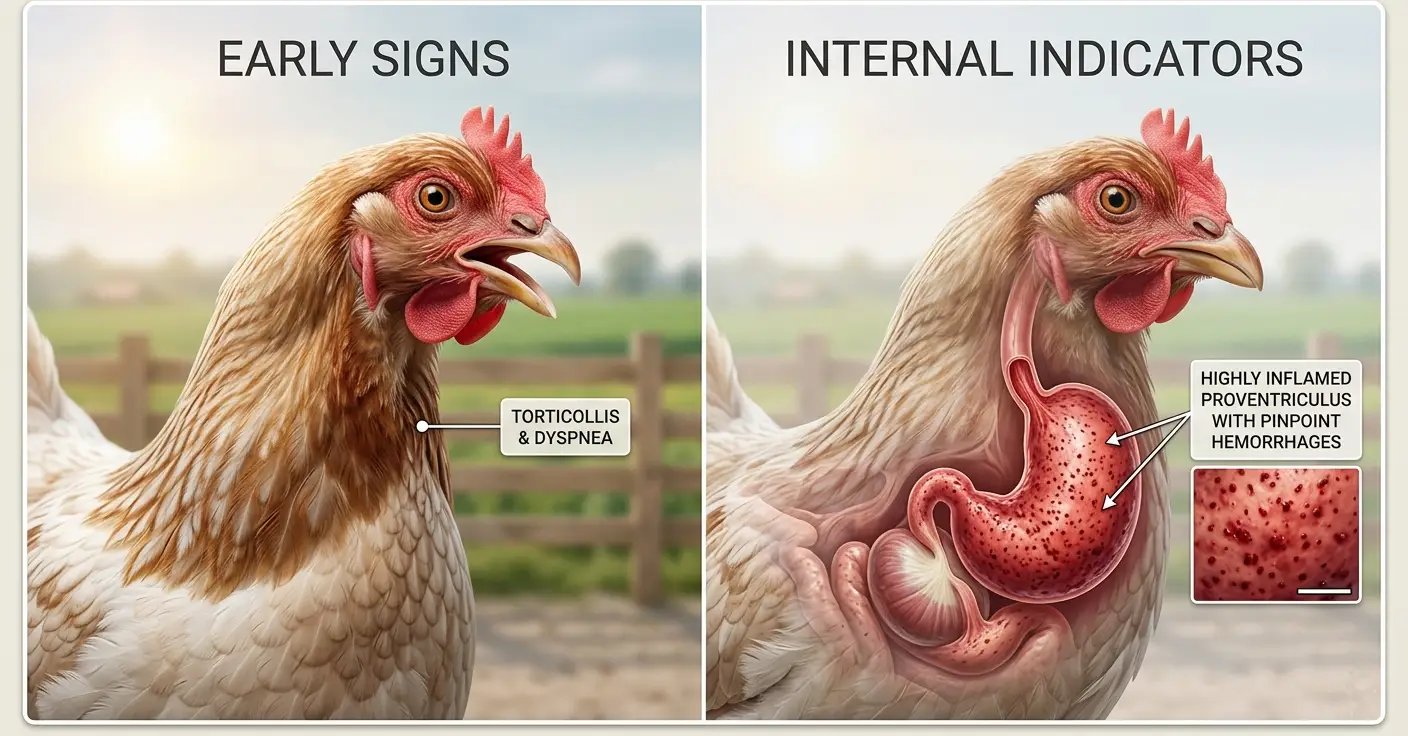
- Velogenic neurotropic (VNND): Primarily attacks the nervous system; causes tremors, torticollis, and paralysis alongside respiratory signs.
Both velogenic subtypes circulate in the West African poultry population. Both produce outbreak mortalities that commercially vaccinated flocks experience when vaccination has failed — which is the critical distinction: vaccination failure, not vaccine absence, drives most commercial outbreak mortality in the region.
How Newcastle Disease Spreads
NDV is shed in respiratory secretions, fecal material, and feather dust. It survives in the environment for variable periods depending on temperature and humidity: 3–7 days on surfaces at 20°C, up to 30 days in cold, humid conditions.
Transmission routes in order of field significance in West Africa:
- Airborne spread from infected backyard or village poultry in the surrounding area — the primary route on farms within 2–3 km of unvaccinated flocks
- Fomite transmission on footwear, clothing, equipment, and vehicles entering the farm
- Wild bird transmission — free-flying birds that have contacted infected village poultry and enter open-sided layer houses
- Live bird market exposure — workers who visit live bird markets and return to the farm the same day
The implication is direct: no biosecurity protocol eliminates Newcastle disease exposure risk in West Africa. The protocol manages and reduces exposure. Vaccination manages and reduces susceptibility. The combination is what prevents an outbreak.
Understanding Gumboro Disease: The Immune System Target
The Pathogen and Its Mechanism
Infectious Bursal Disease Virus (IBDV) is a birnavirus that infects immature B-lymphocytes (precursor B-cells) in the bursa of Fabricius — the primary lymphoid organ responsible for B-cell development and differentiation in birds.
The bursa is located in the dorsal cloaca and is most active between weeks 2 and 8 of a chick’s life. During this period, it produces B-lymphocytes that will populate the spleen and lymph nodes as mature antibody-producing cells. When IBDV destroys the bursal lymphoid tissue during this developmental window, B-cell production is permanently reduced for that production cycle.
The damage is cumulative and non-reversible: a chick that loses 60% of its bursal lymphoid tissue at week 3 to a Gumboro outbreak will have 60% of the humoral immune capacity of an unaffected bird for the rest of its life. It cannot respond normally to the Newcastle vaccination at week 7. It cannot respond normally to any subsequent antigenic challenge. It is permanently immunocompromised.
Subclinical vs. Clinical Gumboro
Clinical IBD presents as an acute outbreak with visible mortality (1–20%), wet droppings, huddling, ruffled feathers, and prostration in affected birds. It is relatively easy to diagnose and triggers immediate investigation.
Subclinical IBD — caused by variant IBDV strains or by infection of birds with partial maternal antibody protection — produces no visible acute signs. Mortality does not rise. The flock appears normal. The bursa is being silently destroyed. The first indication of subclinical IBD is often the finding at necropsy that bursal atrophy is present in birds dying from an unrelated cause — or, more damagingly, the finding that vaccination responses in the rearing period are consistently weaker than expected.
Subclinical Gumboro is significantly more prevalent and significantly more economically damaging than clinical Gumboro in well-vaccinated commercial flocks in West and Central Africa. It goes largely undetected because it does not produce a crisis event.
Why Outbreaks Happen in Vaccinated Flocks: The Three Failure Categories
The most important question for a commercial layer farmer is not “how do I vaccinate against Newcastle and Gumboro?” — most farmers know the answer to that. The most important question is “why do outbreaks occur in flocks that were vaccinated?” — because that is where the actual problem lies.
Failure Category 1: Vaccination Timing Errors (Maternal Antibody Interference)
Day-old chicks carry passive immunity transferred from the hen through the yolk — maternal antibodies that provide early protection but also neutralize live vaccines administered too early. A La Sota NDV vaccine given to a chick at day 7 with high maternal antibody titers against NDV is neutralized before it can replicate and stimulate active immunity. The vaccination event occurred. No protective response was produced.
The critical timing error with Gumboro is even more damaging. IBD maternal antibody titers vary significantly between individuals in the same flock — some chicks have high titers (and the vaccine is neutralized), some have moderate titers (and the vaccine is partially effective), and some have low titers (and the vaccine replicates, but the bird is also susceptible to field challenge before immunity develops). This variance creates the immunological gap: the window during which some birds have lost maternal protection but have not yet developed vaccine-induced immunity.
The solution to timing failure: Maternal antibody mapping — ELISA testing of 20–30 day-old chicks from each incoming flock batch to determine the actual maternal antibody titer distribution. This data allows vaccination timing to be calibrated to the actual titer profile rather than a standardized schedule that assumes average titers.
Where maternal antibody mapping is not available, use the following practical heuristics for West African commercial layer operations:
- First NDV vaccination: day 10–14 (not day 7, which is too early for most flocks sourced from standard commercial hatcheries in the region)
- First Gumboro vaccination: day 14–18, using an intermediate strain vaccine
- If Gumboro history on the farm suggests high maternal titers (Gumboro outbreaks despite standard vaccination in previous flocks): use an intermediate-plus strain (D78, Bursine-2 Plus) at day 16–18, followed by a booster at day 24–28
Failure Category 2: Cold Chain Breakdown
A vaccine that was manufactured correctly, packaged correctly, and labeled for the correct storage temperature delivers zero protective immunity if it is stored or transported above its specified temperature range before administration.
Live attenuated vaccines — La Sota, Clone 30, IBD intermediate strains — are freeze-dried biological products that must be maintained at 2–8°C from the point of manufacture to the point of reconstitution. Temperatures above 8°C during storage or transport cause progressive viral inactivation. There is no visual indicator that the vaccine has been inactivated — the reconstituted product looks identical to an effective vaccine. The flock receives the vaccination. No immunity is produced.
Cold chain failure points in West African commercial layer production:
- Vaccines purchased from veterinary stockists who do not use temperature-monitored refrigeration
- Vaccines transported to the farm in a bag at ambient temperature (30–38°C) rather than in an insulated cooler with ice packs
- Vaccines removed from refrigeration more than 2 hours before use
- Vaccines stored in a shared farm refrigerator that is opened frequently and runs at 12–18°C rather than the required 2–8°C
- Vaccines that have passed their expiry date but were not discarded because “they looked fine”
Cold chain protocol that eliminates most of these failures:
- Purchase vaccines only from suppliers who store them in dedicated pharmaceutical refrigerators with temperature logs available on request
- Transport in an insulated cooler with ice packs — not loose ice, which creates pools of 0°C water that can freeze vaccines if they make direct contact
- Remove from the cooler only at the moment of reconstitution
- Use within 1–2 hours of reconstitution — discard unused reconstituted vaccine
- Record vaccine batch number, manufacture date, expiry date, storage temperature log, and name of vaccine administrator for every event
Failure Category 3: Biosecurity Failure Overwhelming Vaccine-Induced Immunity
Vaccine-induced immunity has a threshold. Below a certain challenge dose of the field virus, a vaccinated bird’s immune response neutralizes the pathogen before clinical disease develops. Above that threshold, even a correctly vaccinated bird can be infected — the response is delayed, or insufficient relative to the pathogen load, and the virus replicates enough to cause disease before immune clearance occurs.
High biosecurity environments reduce the challenge dose. Low biosecurity environments increase it. A farm with open visitor access, no footbath discipline, proximity to live bird markets, and mixed-age flocks sharing equipment and workers is presenting its birds with a continuous, high-dose field virus challenge that vaccine-induced immunity cannot reliably overcome.
This is why two farms with identical vaccination programs — one with strong biosecurity, one without — have dramatically different outbreak rates. The vaccine is the same. The challenge environment is not.
Biosecurity: The Non-Negotiable Foundation
Vaccination without biosecurity is fire insurance without smoke detectors. The policy pays when the fire occurs, but the fire should have been prevented.
The Biosecurity Hierarchy for Newcastle and Gumboro Prevention
Level 1 — Farm perimeter (keep the virus out):
- Controlled access point: A single entry gate with a staffed checkpoint. All visitors log in, change into farm-provided footwear, and pass through a footbath before entering the production area. The footbath must contain active disinfectant — 2% formalin, 4% sodium hypochlorite, or a commercial quaternary ammonium compound at label concentration — and must be refreshed daily. A footbath containing dirty water or expired disinfectant is not a biosecurity measure.
- Vehicle disinfection: Vehicles that enter the farm perimeter — feed trucks, egg collection vehicles, veterinary vehicles — should pass through a wheel dip or receive a spray disinfection of tires and wheel wells before entering. Newcastle disease virus can be transported on vehicle tires from infected farms and live bird markets.
- No visitors from live bird markets on the same day: NDV can be carried on the clothing and footwear of workers who have visited live bird markets. The minimum buffer is 48 hours between a live bird market visit and farm entry — 72 hours during outbreak periods. This policy must apply to the farmer as much as to farm workers.
Level 2 — Between houses (contain what enters):
- Separate equipment per house: Feed scoops, egg collection trays, litter tools, and vaccination equipment should not move between houses. NDV and IBDV can be carried on fomites between houses on equipment that contacts fecal material or respiratory secretions.
- Clothing change between houses: Farm workers who move between a sick or suspected house and healthy houses before changing and disinfecting transfer whatever pathogen is in the first house to the second.
- Dead bird disposal: Dead birds should be collected in sealed bags and removed from the house immediately — not stored at the house end for daily collection. Dead birds shed virus at high concentration and are a primary intra-house contamination source.
Level 3 — All-in/all-out management:
Operating multiple age groups on the same farm — with different flocks at different stages of the production cycle — creates a continuous pathogen cycle. Older birds that have field-strain immunity shed virus that challenges younger, partially immune birds. Younger birds that develop new infections spread them to other houses.
All-in/all-out management — where the entire farm is restocked at the same time and depopulated at the same time, with a complete cleanout and disinfection between cycles — breaks the virus cycle at the farm level. It is the single most effective structural biosecurity measure available and is non-negotiable for farms with a history of recurrent Newcastle or Gumboro outbreaks.
House Cleanout and Disinfection Protocol Between Flocks
The disinfection interval between flocks is the only period during which the pathogen load in the house environment can be reduced to near-zero. The sequence matters:
- Remove all birds, litter, and feed residues completely. IBDV in particular is extremely environmentally resistant — it can survive in dried litter and poultry dust for months at ambient temperature. Litter removal eliminates the primary environmental reservoir.
- Dry clean all surfaces — walls, cage structures, feeding and watering equipment — removing all organic material before applying disinfectant. Disinfectants do not penetrate organic matter effectively. Any surface covered in fecal material or feed residue is not being disinfected, regardless of what chemical is applied to it.
- Wash with pressurized water (hot water where available). Hot water above 60°C inactivates both NDV and IBDV.
- Apply disinfectant at the label concentration. For Newcastle disease: quaternary ammonium compounds, aldehydes (formalin), and oxidizing agents (hydrogen peroxide, peracetic acid) are all effective. For Gumboro: IBDV is non-enveloped and significantly more resistant to chemical disinfection than NDV. Use formaldehyde (formalin fumigation at 30–40 mL/m³ of space, or formalin spray at 2% concentration) for reliable IBDV inactivation.
- Allow the disinfectant adequate contact time — minimum 30 minutes for spray applications. Disinfectant applied and immediately wiped or rinsed has not completed its killing action.
- Rest period of a minimum of 2 weeks between disinfection and restocking. This is not a management preference — it is the period during which any surviving environmental virus (particularly IBDV) continues to decay without the biological amplification provided by a live host.
Outbreak Recognition: What Newcastle and Gumboro Look Like
Speed of recognition determines speed of response. The speed of response determines how many birds and how much production can be saved.
Newcastle Disease — Clinical Recognition
Early signs (days 1–3 of outbreak):
- Sudden increase in respiratory distress — rattling, coughing, labored breathing, audible when walking through the house during quiet periods
- Elevated feed and water intake depression beyond what heat or normal variation explains
- Initial mortality spike — 0.5–2% within the first 48 hours in velogenic outbreaks
- Green or watery diarrhea appearing in multiple birds simultaneously
Advanced signs (days 3–7):
- Torticollis (twisted neck), circling, or one-sided paralysis — nervous system signs from velogenic neurotropic strains
- Sudden mortality escalation — 5–15% per day in unvaccinated or inadequately vaccinated flocks
- Dramatic egg production collapse — 30–80% drop within 3–5 days
- Shell quality failure — soft-shelled, rough-shelled, or shell-less eggs from previously well-shelling birds
Gross pathology at necropsy:
- Hemorrhagic lesions in the proventriculus (the glandular stomach) and along the intestinal mucosal surface — the hallmark of velogenic viscerotropic NDV
- Tracheal hemorrhage and congestion
- Enlarged, dark spleen
A provisional diagnosis of velogenic Newcastle disease can be made on the combination of clinical signs + characteristic proventricular hemorrhage at necropsy. Confirm by submitting fresh tissue samples (trachea, brain, spleen, intestine) in 10% formalin for histopathology and in virus transport medium at 4°C for PCR or virus isolation.
Gumboro Disease — Clinical Recognition
Acute clinical Gumboro (week 2–5 of life):
- Affected birds huddling, with ruffled feathers, and reluctant to stand
- Watery, whitish diarrhea — soiling of the vent feathers
- Pecking at their own vent (vent irritation from diarrhea)
- Mortality peaking at 3–5 days post-infection, declining over 7–10 days total
- House-wide depression — even apparently unaffected birds appear lethargic
Gross pathology at necropsy:
- Bursa of Fabricius enlarged, edematous, and cream-colored in acute infection; atrophied and smaller than normal in post-acute stages
- Edema and hemorrhage in the bursal tissue
- Muscle hemorrhage — particularly in the breast muscles and inner thighs (petechiae and ecchymoses)
- Kidneys enlarged, pale, and showing urate deposition
Subclinical Gumboro:
- No visible mortality event
- Bursal atrophy found incidentally at necropsy of dead birds
- Weaker-than-expected serological responses to subsequent vaccinations
- Increased susceptibility to respiratory and enteric secondary infections
Submit fresh bursal tissue in virus transport medium for PCR confirmation. Bursal tissue in formalin for histopathology to characterize the degree of lymphoid depletion.

Emergency Response When an Outbreak Occurs
Recognition alone does not stop an outbreak. The response in the first 24–48 hours determines the difference between a contained event and a flock-wide catastrophe.
Immediate Actions (First 24 Hours)
Step 1 — Isolate the house. Lock the affected house. No workers, equipment, or materials enter or exit without full protective gear (disposable coveralls, boot covers, gloves) and disinfection on exit. The house is now a contaminated zone.
Step 2 — Implement emergency biosecurity across the farm. Suspend all non-essential movement between houses. Disinfect all footwear and hands before and after every contact with birds. Alert all workers to the outbreak status.
Step 3 — Contact your veterinarian immediately. Both Newcastle disease and Gumboro are notifiable in most countries, including Cameroon and Nigeria. Beyond the legal obligation, a veterinarian is needed to confirm the diagnosis, guide treatment (supportive care), and manage the vaccination response in adjacent houses.
Step 4 — Emergency vaccination of adjacent healthy houses. In a confirmed Newcastle outbreak on-farm, immediately administer a live NDV vaccine (La Sota or Clone 30) by spray or eye drop to all healthy houses on the farm. Vaccinating during an outbreak does not cure infected birds — it raises the immune status of exposed but not yet infected birds and can reduce mortality and accelerate recovery in partially immune birds.
Step 5 — Supportive care for affected birds. There is no specific antiviral treatment for NDV or IBDV. Supportive care during the acute phase:
- Vitamin and electrolyte supplementation in drinking water — oral electrolytes + vitamins A, D, E, and C — to support immune function and reduce secondary bacterial infection risk
- Broad-spectrum antibiotic cover (oxytetracycline, amoxicillin) for secondary E. coli and Mycoplasma infections that exploit the immune compromise from the primary viral disease
- Ensure water and feed remain accessible — heat-stressed or neurologically affected birds may require feeder height adjustment
Step 6 — Record everything. Daily mortality count by house. Production data. Water and feed consumption. Clinical progression. This record is needed for insurance claims, for the post-outbreak investigation, and for designing the next flock’s vaccination program.
Post-Outbreak Investigation
After the outbreak is resolved, investigate how it occurred before restocking. The questions that need answers:
- Were vaccine batch numbers and expiry dates recorded for the last vaccination event? Obtain laboratory viability testing of the same batch if still available.
- What was the cold chain history of the vaccines used?
- When was the last vaccination event before the outbreak, and what was the interval?
- Was the vaccination administered correctly — correct dilution, correct route, confirmed consumption in drinking water vaccination?
- What was the biosecurity status of the farm in the 14 days before the outbreak — visitor log, live bird market exposure, new equipment or vehicles on-farm?
Every outbreak has a traceable cause. Finding it prevents the next one.
Monitoring System: Active Surveillance Before the Crisis
The farms that prevent outbreaks rather than respond to them are the farms that run active monitoring programs — not waiting for mortality to signal a problem, but looking for early indicators that disease challenge is increasing or immunity is declining.
Weekly Flock Health Observation
Designate a specific time — the same time each day, before the lights go out — for a structured flock health walk-through. The observer walks the full length of the house, listening for respiratory sounds, observing posture and activity level, and counting the number of birds that do not stand or move normally. Record the count. Any increase in the count from the previous week is an early warning.
A flock health walk-through takes 10–15 minutes per house. It is the cheapest early warning system available and the one most consistently skipped by operations that claim they do not have time.
Mortality Trend Monitoring
Plot daily mortality count — not percentage, but absolute count — on a weekly graph. A flock running 2–3 birds per day mortality at week 20 that jumps to 8–10 birds per day on day 144 without explanation has a problem that will be visible in the mortality graph before it is visible in the production data.
Set a mortality threshold: if daily mortality exceeds 0.1% of the flock on any single day, investigate that day. Do not wait to see if it was an anomaly. It may have been. It may also have been the first day of a Newcastle disease outbreak.
Serological Monitoring
Conduct serology on a representative flock sample 4 weeks after each vaccination event to confirm that protective immunity has been established:
- Newcastle disease HI titration: Sample 20 birds per house. Target: geometric mean titer (GMT) above 4 log₂ (equivalent to HI titer ≥ 1:16) with fewer than 20% of birds below 3 log₂. A flock with GMT below 4 log₂ at 4 weeks post-vaccination is not adequately protected — investigate vaccination failure and revaccinate.
- IBD ELISA: Sample 20 birds per house. Evaluate both the mean titer and the coefficient of variation (CV). High CV — wide variation between individual birds — indicates non-uniform seroconversion, which is the characteristic serological signature of a vaccination event during which some birds were still under maternal antibody influence, and others were not.
Summary
Newcastle disease and Gumboro remain the most economically destructive infectious diseases in West and Central African layer production, not because vaccines do not exist, but because vaccination programs fail at three specific and correctable points: timing calibrated to maternal antibody profiles that are not measured, cold chains that break without detection, and biosecurity environments that overwhelm the immunity vaccines produce.
Closing those three gaps — through maternal antibody-guided vaccination timing, cold chain discipline at every step, and biosecurity protocols that treat the farm as a biological containment system — is what converts “vaccine-preventable disease” from a theoretical category to an operational outcome.
The vaccines work. The cold chain must work. The biosecurity must work. The monitoring must catch what fails before the mortality does.
Every outbreak in a vaccinated flock is an investigation report waiting to be written. Write it before the next flock arrives.